The vector that we use is a small virus known as adeno-associated virus (AAV), which is non-pathogenic (i.e. carrier) of the normal gene that can safely bring the normal RPGR genes back into the retinal cells without harming them. 
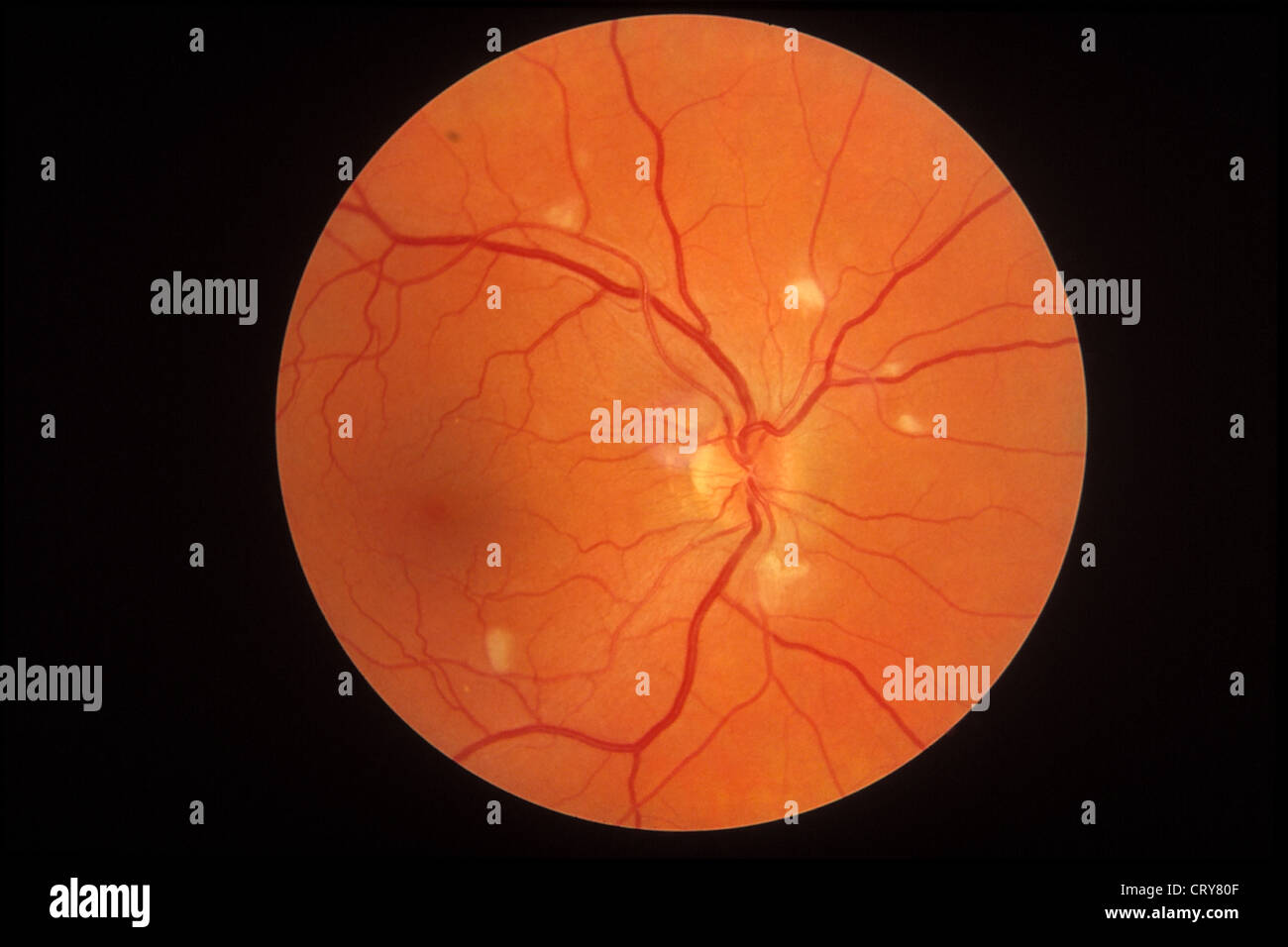
The new technique involves putting normal copies of the affected RPGR gene back into the cells of the retina to help them to function normally. In order to do this, we need to use a vector (i.e. There are currently no effective treatments available for XLRP, but we have developed a new technique of gene therapy which we believe may help to slow or even stop the degeneration. loss of night vision) in adolescence, followed by a gradual loss of peripheral vision which results in progressively worsening ‘tunnel vision’. Ultimately, central vision is lost by the fourth decade, with m ost XLRP patients becoming legally blind by the age of 40. Sight loss in XLRP begins with ‘night blindness’ (i.e. The absence of RPGR in the retinal cells causes them to die over time, resulting in a progressive degeneration of the retina and consequent loss of vision. The RPGR gene encodes a special protein called X-linked retinitis pigmentosa GTPase regulator (RPGR) which plays a key role in the development of the cells making up the retina, which is the light-sensitive layer (like a camera film) that lines the back of the eye. 
Men, however, only have one X-chromosome. Women have two X-chromosomes and so a normal RPGR gene on one X-chromosome can compensate for a defective RPGR gene on the other X-chromosome to some extent. The disease is caused by a defect in the RPGR gene which is located on the X-chromosome, and this is why the disease affects men and women differently. X-linked retinitis pigmentosa (XLRP) is an incurable genetic disease that causes blindness in men, and affects approximately one in 15,000 people.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
